c1
- Created by: milliebramley
- Created on: 15-02-17 13:48
The limestone cycle

Crude Oil
-
Crude oil is found trapped in some of the sedimentary rocks of the Earth's crust. Millions of years ago, huge numbers of microscopic animals and plants - plankton - died and fell to the bottom of the sea. Their remains were covered by mud. As the mud sediment was buried by more sediment, it started to change into rock, as the temperature and pressure increased. The plant and animal remains were ‘cooked’ by this process, and slowly changed into crude oil. Oil is less dense than the water in the rocks and will rise as a result of pressure from below, often escaping altogether if the rocks are permeable.If some of the rocks above the oil are impermeable, the oil cannot rise through them, so it gets trapped underneath.
- Crude Oil is a mixture of hydrocarbons
- hydrocarbons are molecules which contain hydrogen and carbon atoms only.
- Hydrocarbons have different lengths and therefore different boilng points.
- They can be seperated by fractional distillation.
- The demand for short hydrocarbon molecules is greater than their supply in crude oil, so a reaction called cracking is used. Cracking converts long alkane molecules into shorter alkanes and alkenes, which are more useful.
- The exploitation of oil can damage the environment - for example, through oil spills.
Distillation
- Distillation is a process that can be used to separate a pure liquid from a mixture of liquids. It works when the liquids have different boiling points. Distillation is commonly used to separate ethanol - the alcohol in alcoholic drinks - from water.
- The mixture is heated in a flask. Ethanol has a lower boiling point than water so it evaporates first. The ethanol vapour is then cooled and condensed inside the condenser to form a pure liquid. The thermometer shows the boiling point of the pure ethanol liquid. When all the ethanol has evaporated from the solution, the temperature rises and the water evaporates
.
- This is the sequence of events in distillation:
- heating → evaporating → cooling → condensing
Fractional distillation pt1
Fractional distillation differs from distillation only in that it separates a mixture into a number of different parts, called fractions. A tall column is fitted above the mixture, with several condensers coming off at different heights. The column is hot at the bottom and cool at the top. Substances with high boiling points condense at the bottom, and substances with low boiling points condense at the top. Like distillation, fractional distillation works because the different substances in the mixture have different boiling points.
Fractional distillation of crude oil
Because they have different boiling points, the substances in crude oil can be separated using fractional distillation. The crude oil is evaporated, and its vapours allowed to condense at different temperatures in the fractionating column. Each fraction contains hydrocarbon molecules with a similar number of carbon atoms.
Fractional distillation pt2
.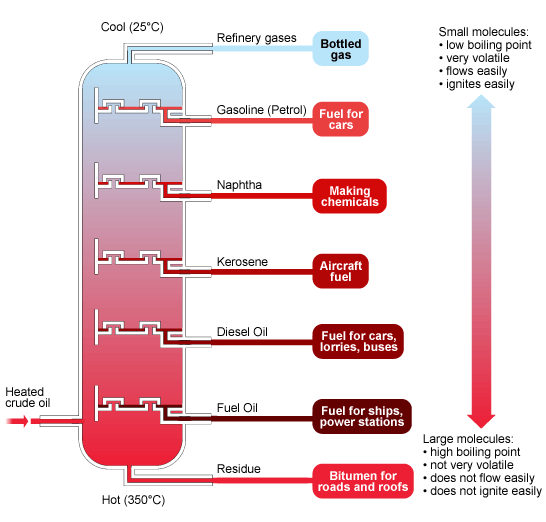 Oil fractions
Oil fractions
The diagram summarises the main fractions from crude oil, their uses, and the trends in properties. Note that the gases condense at the top of the column, the liquids in the middle, while the solids stay at the bottom.
Fractional distillation pt3
Here are the main fractions in list. They are in increasing order from small molecules - gases - to large ones.
- LPG - contains propane and butane, used in bottled cooking gas
- petrol - fuel for cars
- naphtha - used in the chemical industry
- paraffin - aircraft fuels
- heating oil - diesel and heating
- fuel oils - fuel for ships and power stations
- bitumen - surfaces for roads and roofs
Hydrocarbons with small molecules make better fuels than hydrocarbons with large molecules, because they are volatile, flow easily, and are easily ignited.
Problems with exploiting oil pt1
Drilling for oil
Geologists can often tell where oil is trapped by looking at the structure of the rocks. Oil tends to be trapped where rocks are domed upwards, or where permeable rocks are in contact with impermeable rocks at a fault line.
Oil companies can drill down through the impermeable rocks to get it out. They are then able to turn the oil into products we can use.
Crude oil takes millions of years to form, and we are using it up more quickly than it is created. Present estimates suggest world supplies of crude oil will run out in about 30 years, unless we use it more efficiently. There are additional reserves of oil in rocks called oil shale. However, it is expensive to extract oil from oil shale because it needs to be heated to release the oil.
Problems with exploiting oil pt2
Environmental problems
Oil spills cause a lot of harm to the environment, both at sea and on land
- Oil is carried from oil fields to refineries using ocean-going tankers. If the oil is spilled, it causes considerable damage to the environment:
Oil slicks travel across the sea, far from the original spill.
Beaches and wildlife are harmed when they are coated with oil.
Cracking
Cracking
Fuels made from oil mixtures containing large hydrocarbon molecules are not efficient. They do not flow easily, and are difficult to ignite. Crude oil often contains too many large hydrocarbon molecules, and not enough small hydrocarbon molecules, to meet demand. This is where cracking comes in.
Cracking allows large hydrocarbon molecules to be broken down into smaller alkane and alkene molecules:
- smaller hydrocarbons are more useful as fuels
- alkenes are useful, because they are used to make polymers
Fractions containing large hydrocarbon molecules are vaporised and passed over a hot catalyst. This breaks chemical bonds in the molecules, and forms smaller hydrocarbon molecules.
Cracking is an example of a thermal decomposition reaction.
Links
BBC Science and Nature
The natural world and beyond!
On the web
BBC iD
Search term:
BBC navigaRadio
BBC © 2014 The BBC is not responsible for the content of external sites. Read more.
The reactivity series
Alloys
An alloy is a mixture of two elements, one of which is a metal. Alloys often have properties that are different to the metals they contain. This makes them more useful than the pure metals alone. For example, alloys are often harder than the metal they contain.
Alloys contain atoms of different sizes, which distorts the regular arrangements of atoms. This makes it more difficult for the layers to slide over each other, so alloys are harder than the pure metal.
- iron from a blast furnace contains 96% iron. the impurities make it brittle and so it has liited uses.
- most iron is converted into steel. steels are alloys since they are mixtures of iron with carbon.
- some steels contain other metals
- alloys can be designed to have properties for specific uses. low carbon steels are easily shaped, high carbon steels are hard and stainless steels are resistant to corrosion.


Comments
No comments have yet been made