C4 Chemical patterns
5.0 / 5 based on 1 rating
- Created by: sophiedoherty17
- Created on: 24-02-16 18:13
What does the nucleus contain?
Protons and neutrons.
1 of 35
What charge does the nucleus have? Why?
Positive charge because of the protons.
2 of 35
What is the relative mass and charge of a proton?
Relative mass- 1 Charge- +1
3 of 35
What is the relative mass and charge of a neutron?
Relative mass- 1 Charge- 0
4 of 35
What is the relative mass and charge of an electron?
Relative mass- 0 Charge -1
5 of 35
What does the number of protons in a neutral atom always equal?
The number of electrons.
6 of 35
What is an ion?
A charged atom.
7 of 35
The number of protons in an atom decides what?
What element it is.
8 of 35
What colour flame does lithium produce?
Red
9 of 35
What element produces a yellow flame?
Sodium.
10 of 35
What element produces a purple flame?
Potassium.
11 of 35
The wavelengths emitted by atoms can be recorded as what?
Line spectrums.
12 of 35
What did Newlands organise elements into?
Newlands organised elements into groups of eight in order of increasing proton number.
13 of 35
Why did Newlands periodic table not work?
Newlands didn't leave any gaps for undiscovered elements, he also mixed metals and non-metals and the groups contained elements that didn't have similar properties.
14 of 35
Who organised elements into groups of three?
Dobreiner arranged elements into groups of three with the middle element of each triad having a relative atomic mass that was the average of the other two.
15 of 35
Which scientists came up with the periodic table that we use today? Why did it work?
Mendeleev put the elements in increasing proton order, but left gaps to keep elements with similar properties in the same group. These gaps were for undiscovered elements.
16 of 35
Which side of the periodic table are metals found on?
Metals are found on the left side of the periodic table.
17 of 35
Where are the non-metals found on the periodic table?
Non-metals are found on the right side of the periodic table.
18 of 35
What does the group number tell you?
The number of electrons that an element has in it's outer shell.
19 of 35
How many electrons can the shells of an element hold?
2 electrons in the first shell and 8 in any shells after this.
20 of 35
Why do ions become positively charged when they lose and electron?
When an electron is lost there are then more proton with a positive charge than electrons in the atom, so the overall charge becomes positive.
21 of 35
What is the name of group 1?
The alkali metals.
22 of 35
What happens to the elements as you move down group 1?
The elements become more reactive, have a higher density and lower melting and boiling points.
23 of 35
What elements from group one produce hydrogen when put into water?
Lithium, sodium and potassium.
24 of 35
Alkali metals react with chlorine to produce what?
Salts
25 of 35
What is the name of group 7?
Halogens
26 of 35
What happens as you move down group 7?
The elements become less reactive and have higher melting and boiling points.
27 of 35
What is flourine?
Flourine is a very reactive, poisonous yellow gas at room temperature.
28 of 35
What is chlorine?
Chlorine is a fairly reactive, poisonous dense green gas at room temperature.
29 of 35
What is bromine?
Bromine is a dense, poisonous orange liquid at room temperature and forms an orange gas.
30 of 35
What is iodine?
Iodine is a dark grey solid at room temperature and turns into a purple gas when heated.
31 of 35
The halogens react with alkali metals to form salts called what?
Metal halides.
32 of 35
The halogens react with alkali metals to form coloured solids called what?
Iron halides.
33 of 35
Alkali metals are very reactive so are store in what?
Oil
34 of 35
Halogens have to be used inside a what so that you don't breathe in the fumes.
A fume cupboard.
35 of 35
Other cards in this set
Card 2
Front
What charge does the nucleus have? Why?
Back
Positive charge because of the protons.
Card 3
Front
What is the relative mass and charge of a proton?
Back
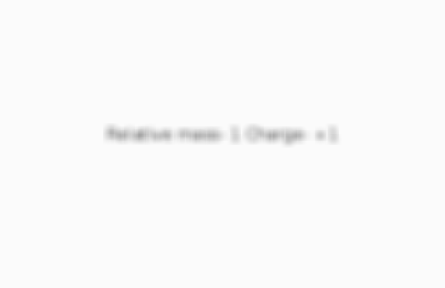
Card 4
Front
What is the relative mass and charge of a neutron?
Back

Card 5
Front
What is the relative mass and charge of an electron?
Back

Related discussions on The Student Room
- Help chemistry urgent »
- Chem alevel help »
- Chemistry Alevel question »
- Chemical structures Biology A level »
- Please help me find marks (2 marks of an A* for A level chemistry) »
- failing chemistry a-level, struggling to revise »
- Why does Scandium only exist in the 3+ ion »
- Cambridge Chemical Engineering Interview »
- Best uni with cheap tuition for chemical engineering »
- chemistry as level equilibria help »
Similar Chemistry resources:
1.5 / 5 based on 4 ratings
0.0 / 5
0.0 / 5
2.0 / 5 based on 1 rating
3.0 / 5 based on 1 rating
0.0 / 5
0.0 / 5
4.5 / 5 based on 11 ratings
2.0 / 5 based on 1 rating
2.0 / 5 based on 1 rating
Comments
No comments have yet been made