Radioactive materials - substances that give our nuclear radiation.
Nuclear radiation - used in medical treatments such as Radiotherapy to cure cancer. - Nuclear radiation can kill the cancer cells - can also kill healthy cells.
Radioactivity - change in structure of radioactive atom and the release of one of the three types of nuclear radiation:
-
- alpha (
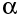 )
)
- beta (
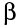 )
)
- gamma (
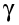 )
)
Nuclear radiation can cause ionisation, producing postively and negatively charged ions when atoms gain or lose electrons. Ionisation can be harmful inside the body - damages cells and can initiate chemical reactions by breaking molecular bonds.
Comments
No comments have yet been made