Chemistry 1
- Created by: chloe
- Created on: 08-05-17 11:55
Atomic Structure
STRUCTURE OF THE ATOM: Protons and Neutrons are found in the nucleus. Electrons orbit the nucleus in shells.
All substances are made of atomsthis is cannot be chemically broken down it is the smallest part of an element. Elements are made of only one type of atom. Compounds contain more than one type of atom. Compounds are held together by bonds. Mixtures contain elements and compounds.
An atom contains equal numbers of protons and electrons. All atoms of an element have the same numberof protons. Atoms of different elements have differentnumbers of protons
Atoms of each element are represented by a chemical symbol e.g. O for oxygen, Na for sodium.
Atomic structure
Periodic Table
Elements in the samegroup in the periodic table
have the samenumber of electrons in their outershell so they have similarchemicalproperties.
E.g. Group 1 Alkali metals
Reactions of group 1 elements with water
Lithium, sodium and potassium all react vigorously with water.
metal + water → metal hydroxide + hydrogen
The metalhydroxides are strong alkalis. The group 1 elements need to be stored under oil to prevent them reacting with oxygen and water vapour in the air.
Each element has its own symbol. Columnsare called groups Elementsin a group have similarproperties. Rowsare called periods. The staircase line splits metals (LEFT) from non-metals (RIGHT)
Elements in Group0 of the periodic table are called the noblegases. They are unreactive because their atoms have stable arrangements of electrons. A full outer shell.
Periodic table
Chemical Reactions
Two types of bonding:
Ionic Bonding: Metaland non-metalreact. Metalsform positive ions, Non-metals form negative ions. Oppositecharges attract. Metals LOSE electrons Non Metals GAIN electrons.
Covalent Bonding:When two non-metals bond. Outermost electrons are shared . A pair of shared electrons forms a bond
Saturated and unsaturated oils
Animal fats -- Satutrated fats contain singele carbons bonds c-c
- solid at room tempature
- Are not good for us
- Increased risk of heart disease
- Increased cholestrol
E.g butter, lard
Vegatable oil -- unsaturated Fats contain double carbon bonds C=C
- liquids at room tmepature
- source of nutrients
- Keeps our artiries clear
- Reduces Heart disease
- lower cholestrol
e.g sunflower oil and olive oil
Saturated and unsaturated oils
Testing for alkenes : You can use bromine water to work out if you have alkenes. bromine water is brown. Alkenes make the bromine go colourless. Akanes do not change the colour of the bromine water- it stays brown
The Earths crust
The earths crust, the atosphere and the oceans are the only sources of mineral and other resources that human need
Atomsphere : most lies within 10 km of the surface, rest os within 100km but is ahrd to judge.
Crust/; solid , 6km benenth oceans 35 benethen land
Mantle : Behaves like a solid can flow very slowly. it is 3000km deep!
Core: made of nickel and iron. outer core is liquid and the inner is solid. raduis is 3500km
Moving contients:
The Earths crust and the upper mantle are cracked into a number of pieces. = tectonic plates . these are constanly moving - just very slowly . motion is caused by convectionc currents in the manlte , due to the heat from radioactive decay.
plate boundaries- The earths crust is spilt into sections called. tectonic plates. Earthquakes and volcaneos happens when plates meet- very difficult to precict.
The Earths crust
Wegener's evidence for continental drift- the same type of fossiliesed animals andf plant are found in the south america and africa. The shape og the east coast of south America fits the west coast of Africa, like pieces in a jigsaw puzzle. Matching rock formations anf mountain chains are found in south America and Africa.
Pangea: If you look at the continents thwy roughly fit together. sciensist think they were once one large land mass called pangea, which then broke off into smaller chunks
The Earths atomsphere
The earth atompshere today: for 200 million years, the proportions gases in the atomsphere simular today
Gas Formular %
Nitrogen N 80
Oxygen O 20
Carbon dioxide CO 0.04
Carbon diocide levels: have increased in the atompshere recently largly to the amount of fossil fuels we burn
The Earths atomsphere - Evolution of the earths at
Phase 1 (1st billion years): = Volcaneo= steam and carbon dioxide
Volcaneos kept erutping giving out steam and carbon dioxide. the early atompshere was nearly only carbon dioxide. the Earth cooled and water vapour condensed to form the oceans
Phase 2- Green plants, bacteria, algae= oxagyen
Green plants, bacteria and algae photosynthiesed ocean. Green plant steadily converted carbon dioxide into oxygen by the process of photosynthesis. Ntirogen released by denitrifying bacteria. plants colonise the land, oxygen levels steadly increased.
Phase 3- ozone layer- animals and humans
The build up of oxygen killed off early organisms- allowing evolution of complex organisms. the oxygen created the ozone layer which blocks harmful UV rays from the sun virtually no carbon dioxide left
Properties and Uses of metal
Transition metals have the following properties:
- Shiney when polished
- Malleable - can be hammerd into shape
- Strong - don't break easily when force is applied
- High melting point
- sonorous- makes a ringing shape when hit
- Ductile- can be stretched into wires
- condict heat and electity
Copper has the properties that make it useful for electrical wiring and plumbing. not very reactive , exellent conductor of electrity, easily bent into shape for water pipes in plumbing.
Periodic table - properties and uses of metal
Used as structual metals to make, buildings, bridges, cars
Crude oil
Crude oil: A mixture of lots of different compounds. Formed from dead sea creatures over millions of years. We separate it into substances with simular boiling points. These called fractions. this is done by the process of fractional distillation
Nearly all compounds of crude oil are hydrocarbons. (hydrogen and carbon only). most of these are alkanes. Alkanes have all single bonds and the general formualur
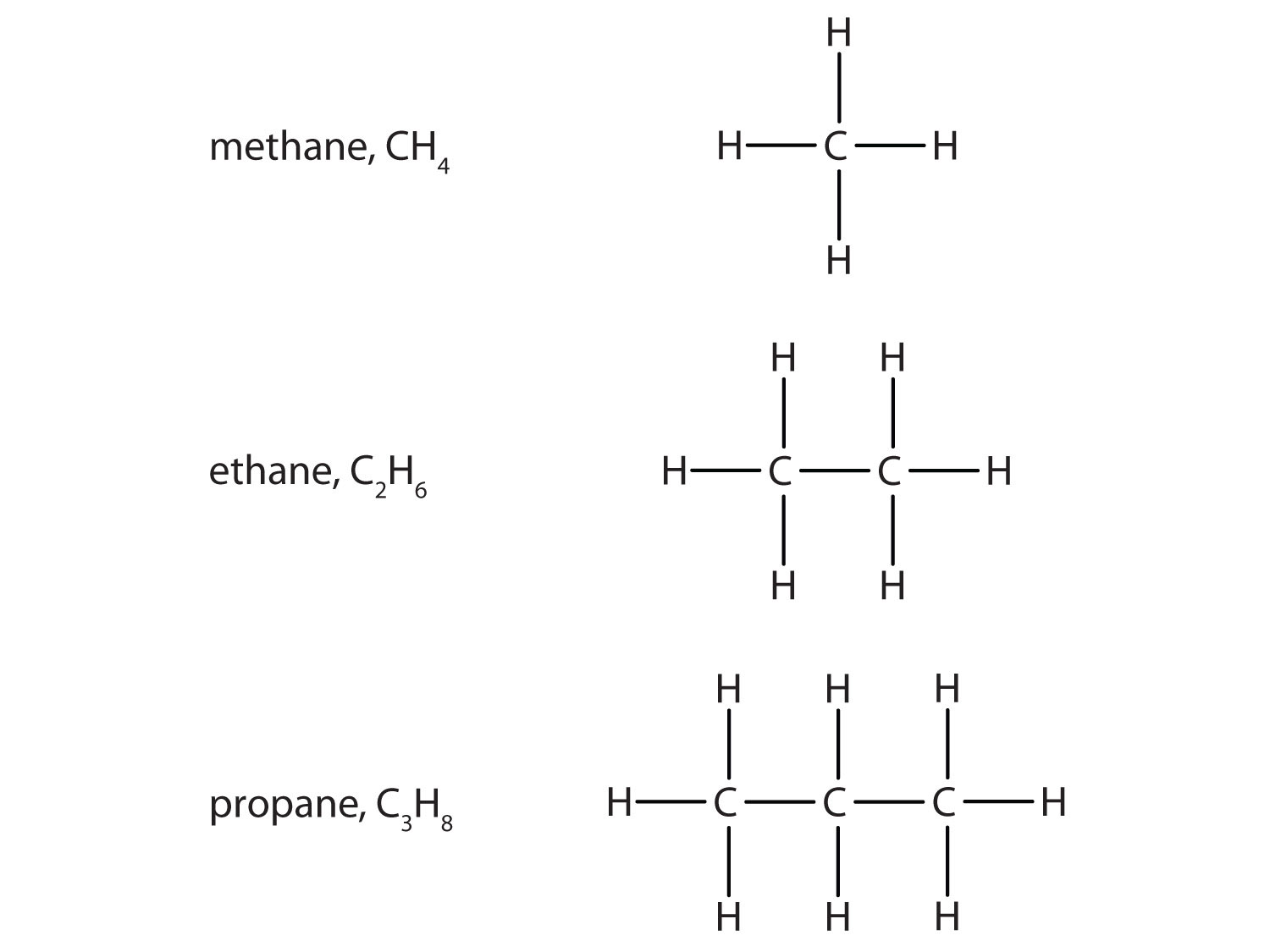
Fractional distillation
Hydrocarbon fuels
Global warming: caused by carbon dioxide , causing the average global tempatures to increase.
Global dimming: caused by particulates relfect sunlight back in to space. not as much loght get to earth.
Sulphur Dioxide gas: is produced by vechials amd power stations when fuel containing sulphur is burned. Sulphur dioxide dissolve in rain and produces acid rain. the sulphur dioxide cam ne removed from waste gases in cars by catalyic converters ans in power stations by reacting with limestones
Hydrogen fuel:
Advanatges- Very clean - no carbon dioxide water is the only product
Disadvantages - hydrogen is explosive, takes up lots of volume = storage become an issue
hydrogen fuel
combustion of hydrocarbons: when burnt in an adequate supply of air alkanes react to firm carbon dioxide E.g
propane + oxygen = carbon dioxide + water
wehn burnt not enough exygen carbon monoxide is formed
Propane + oxygen = carbon monoxide + water
fossil fuels also produce a number of impurties when they are burnt, main pollutants are summaried below
Hydrocarbon fuels
Sulphur dioxide-
- poisonoius gas as it is acidic , causes acid rain, causes engine corrolsion
Nitrogen oxide
- poisonous, triggers asthma attacks, causes acid rain
Particulates
- tiney solid particles, contain carbon un burnt hydrocarbon, carried by air, damages cells in lungs
Ethanol fuel:
Advatages- easily made by ferdermenting sugar canes, gives off carbon dioxide but sugar canes it comes from aborbs carbon dioxide when growing
Disadvanatges- large area of farmland required, less food produced as people used it from fuel instead
Biodiesel
advanatages
- less harful to animals
- particulates reduced
- carbon dioxide neutrals- plants grown to create, it absorbs the same amount of carbon dioxide generates when burnt
Disadvanges
- large areas of farmland is required,
- less food is produced - famine
- destruction of habiats
- freezes a low temps
obtaining useful sunstances from cude oil
mor demand for shorter chain hydrocasrbons. short chain prefferd so long chains "cracked" to make shorter ones
Cracking- this is when a large alkanes is turned into a smaller alkanes and an alkene.
you can crack it into smalle more useful hydrocarbons
hexane= butane + ethnene
alkenes- alkenes are hydrocarbons ( made up of carbon and hydrogen) with carbon- carbon double bonds (c=c). they have the genral formular ( 2 hydrogens for every carbon)
polymers
alkene can be used to make polymers such poly(ethene) and poly(propene. in these reactions may small alkane molecules (monomers) join together to form very laffeg molecules (polymers). this happens at very high pressures and tempatures
Smart polymers- their properies changed by light, tmepatures or other chnages in their surroundings
light snetive plasters- top layer of plasted peeled back, lowet layer exposed to light, adhesive light stickness peels easily off the skin
Hydrogels- have cross- linking chains that traps water act as wound dressing. lets body heal, good for burns
Shape-memory- wound is stitched loosly. temp of the body makes the thread tighten. closes up the wound
polymers
alkene can be used to make polymers such poly(ethene) and poly(propene. in these reactions may small alkane molecules (monomers) join together to form very laffeg molecules (polymers). this happens at very high pressures and tempatures
Smart polymers- their properies changed by light, tmepatures or other chnages in their surroundings
light snetive plasters- top layer of plasted peeled back, lowet layer exposed to light, adhesive light stickness peels easily off the skin
Hydrogels- have cross- linking chains that traps water act as wound dressing. lets body heal, good for burns
Shape-memory- wound is stitched loosly. temp of the body makes the thread tighten. closes up the wound
polymers
Biodegradable plastics : plastics that break down easily. corn strach are built into the plastic. microorganisms is soil feed on corn starch. this breaks down plastic
issue with polymers
biodragdable - farmers sell crops like corn to make plastics, demand for food geos up, food prcies goes up - non -biogradable - don't break down, litter, harm wildlife , alst 100s of years, fill up landfill sites.
polythene- plastic bags and bottles
Polypropene- crate and ropes
Polychloroethene- water pipes and insultation on electricty cable.
calcium carbonate - limetstone
We use limewater to test for carbon dioxide, it turns cloudy
Limewater is made mainly of calcium carbonate. formed from the remains of sea animals that lived millions of years ago. limestone is quarrid and used as building materials. It is also used in chemical industry and for cosmetics.
cement: made by heatuing limestone with clay in a klin.
Mortar- made by mixing cement and sand with water.
concrete- Made by mixingcrushed rocks or stones (called aggregate), cement and sand with water.
Heating limestone- breaking down of chemical by heating is called thermal decomposition.
Calcuim hydroxide: is used to neutralise acidic soils.
The carbonates of magnesuim, copper, zinc, calcium and soduim decompostion on heating in a simular way
Quarrying
Advanatges
- provides jobs
- leads to improvemnt roads
- Filled in to make fishing
- when empty uses as landfill sites
Disadvanatges
- Destroys habitats
- increased emissions
- Noisy and dusty
- Busier roads
Extracting metals
A metal compound within a rock is an ore. The metal is often combined withoxygen. ore are mined and then purified.
Low grade copper ore: small amount of copper
1. phytomining- p;amst aborb copper ions from low-grade ore. plant are burned. copper ions dissolve by adding acid- uise displacment or electorlysis to extract pure copper
2. biobleaching- : bacteria feed on low- grade ore. produce a waste products that contain copper ions. use displacement or electrolysis to extract pure copper
During electrolysis: in solution or moltern compound when electricty is passed through it postive metal ions move towards the negative electrode. Negative non metals ion move towards the positive electrode.
Extracting metals
Copper rich ores: Large amounts of copper.
1. smelting: 80% of copper is produced this way. heating copper ore in a furnace with air. The use electrolysis to purify the copper. Expensive as need lots of heat and power
2. copper sulphate: add sulphic acid to a copper ore. prodcues copper sulphate. extract copper using electrolysis or displacement
The reactivity of a metal determinds the method of extraction. metals above carbon must be extracted from electrolysis, metals below carbon must be extracted by reduction using carbon, coke, or charcoal. Gold and silver ions move towards the postive electrode.
Extracting Metals
Aluminuim
properties- light, low density, oxide layer on the surface prevent corrosion, improved hardness by forming alloys. these alloys are stronger and rigid than a pure aluminuim.
uses: drink cans, cooking oil, sauscepan, overhead cables, airplanes
Extraction- alumium ore is extracted from mined. aluimuim oxide is melted
Titanuim
Properties- strong, oxide layer on the surface prevents corriosn, high melthing point, less dense than most metals
Uses: hip replacement, racing bikes, jet engines, parts of nuclear reactors
Extraction- use soduim and potassium to displace titanuim from it ore. Expensive
Alloys
A metal mixed with other elements is called alloys. alloys are harder than pure metals
Extracting iron-
iron ore goes into the blast furnace and the iron is removed are called reduction reactions. iron is a blast furnace contian about 96% iron. The impurities make it brittle and so it has limited uses
pure metals- regular patterns layer side easily over each other.
Alloy- other elements disrupts regular pattern layer do not slide easily over each other
Alloys
Iron alloys
Steel- iron with carbon and/ or other elements. impureites make it brittle. there are bumber of types of steel alloys: low carbon steel- easily shaped, high carbon steel- very hard, stainless steel- resistant to corrosion
Aluminiuim Alloys
Aluminiium naturally soft mixed with wide range of other elements. all have very different properties E.g aircrafts ir armour plating
Ethanol
Ethanol can be produced by the hydration of ethene with steam in the presence of a catalyst. Or by fermentation with yeast. it is a flammable colourless liquid Boiling point 78 C
Ethanol can be made by fermentiation
Sugar + Yeast = Ethanol + carbon dioxide
Ethanol can also be made by hydration (adding water to) ethene
Ethene + Steam = Ethanol
Uses of Ethanol
- Alcoholic drinks
- fuel
- solvents
- deodorants
- medicine
- perfume
Ethanol
Fermentation
- Uses corn, sugar cane (renewable resource)
- Is a batch process. which needs a lot of workers
- Produces impure ethenol, purified by distillation
- needs a tempature of 30-40 C
- Is a slow reaction
Hydration
- Uses crude oil, which is non -renewable resource
- Is a continious process so is less labour intensive
- Produces pure ethanol
- Needs a tempature if 300 C and high pressure
- Is a fast reaction
Vegetable Oil
Some fruits, seeds, and nuts are rich in oils that can be extracted
Vegetable oil is an important foods:- Provide important nutrients (e.g contain lots of energy = can be used as fuels)
Unsaturated oil contain double bonds (C=C) = they decolourise Bromine water
Benefits of cooking with oil
- Oil has higher boiling point than water
- food cooks quicker
- Outside become crispy
- Inside become softer
- Food absorbs some of the oil
- Higher energy content
- Too much is unhealthy
Vegetable Oil
Extracting Oils
Pressing
- Farmers collect seeds from plants
- seeds are crushed and pressed, then the oil extracted
- Impurities are removed
- Oil us processed to make it into useful product
Distillation
- plants are put into water and boiled
- oils and water evapourate together
- Oil is collected as the liquids separate
Emulsions
Emulsifiers
Stop water and oil separtating into layers. improved texture and taste of foods containing fats and oils. make them more palatable (tasty) and tempting to eat!
Oils - do not dissolve in water, they are immiscible. they don't mix and form layers.
Emulsions- Where oil anf water dispersed (spread out) in each other. they have special propeties.
A food additive- is a substance that is added to a food to improve it's taste, preserve it or change colour. All food additives in our food have E numbers to prove that they have passed safety standards
Emulsifiers have E numbers that begin in 4
Emulsifiers stop oil and water based substances from separting. Emulsifiers are needed in chocolate, mayonise and ice cream
Emulsions
Emulsifiers
Stop water and oil separtating into layers. improved texture and taste of foods containing fats and oils. make them more palatable (tasty) and tempting to eat!
Oils - do not dissolve in water, they are immiscible. they don't mix and form layers.
Emulsions- Where oil anf water dispersed (spread out) in each other. they have special propeties.
A food additive- is a substance that is added to a food to improve it's taste, preserve it or change colour. All food additives in our food have E numbers to prove that they have passed safety standards
Emulsifiers have E numbers that begin in 4
Emulsifiers stop oil and water based substances from separting. Emulsifiers are needed in chocolate, mayonise and ice cream
Related discussions on The Student Room
- Is my degree unusually difficult or am I bust? »
- GCSE paper 1 and paper 2 »
- Chemistry 2023 »
- 10 weeks going from C to A* - Alevel »
- GYG - GCSEs in a month »
- in need of urgent advice, would appreciate any help :) »
- Grade Growth Chronicles | From C's to A's (23-24) »
- GCSE Diary »
- BTEC Level 3 Applied Science Unit 1 May 2022 Exams »
- Revision Struggles?! Join the 2023 TSR All Day Revision Thread! »

Comments
No comments have yet been made