CHEMISTRY 1
- Created by: milliebramley
- Created on: 14-02-17 19:48
elements in the periodic table
- All substances are made of atoms.
- A substance that is made of only one sort of atom is called an element.
- There are about 100 different elements which are shown on the periodic table.
- Elements in the same group in the periodic table have the same number of electrons in their highest energy level (outer electrons which gives them similar chemical properties).
- The elements in group 0 of the periodic table are called Noble gases.
- Noble gases are unreactive because their atoms have stable arrangements of electrons (eight electrons in their outer energy level- except for helium which only had two electrons).
Reactions of Group 1 elements with Water& Oxygen
- Lithium, sodium and potassium are easily cut with a blade. The freshly cut surfaces are silvery and shiny, but quickly turn dull as the metal reacts with oxygen in the air. The group 1 metals react vigorously with oxygen to form metal oxides. Lithium burns with a red flame, sodium with a yellow-orange flame, and potassium burns with a lilac flame.
- Lithium, sodium and potassium all react vigorously with water to form a metal hydroxide and hydrogen:
metal + water → metal hydroxide + hydrogen
The metal hydroxides are strong alkalis.
The group 1 elements need to be stored under oil to prevent them reacting with oxygen and water vapour in the air.
ions
Ions are electrically charged particles formed when atoms lose or gain electrons. They have the same electronic structures as noble gases. Metal atoms form positive ions, while non-metal atoms form negative ions. The strong electrostatic forces of attraction between oppositely charged ions are called ionic bonds. Ionic compounds have high melting and boiling points.
- Ions are electrically charged particles formed when atoms lose or gain electrons. This loss or gain leaves a complete highest energy level, so the electronic structure of an ion is the same as that of a noble gas - such as a helium, neon or argon.
Metal atoms and non-metal atoms go in opposite directions when they ionise:
- Metal atoms lose the electron, or electrons, in their highest energy level and become positively charged ions.
- Non-metal atoms gain an electron, or electrons, from another atom to become negatively charged ions.
How many charges?
How many charges?
There is a quick way to work out what the charge on an ion should be:
- The number of charges on an ion formed by a metal is equal to the group number of the metal
The number of charges on an ion formed by a non-metal is equal to the group number minus eight
hydrogen forms H+ ions
Metal Ions
Metal ions
You need to be able to show the electronic structure of some common metal ions, using diagrams like these:
Lithium, Li
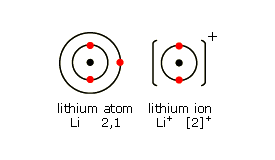 Lithium is in Group 1. It has one electron in its highest energy level. When this electron is lost, a lithium ion Li+ is formed.
Lithium is in Group 1. It has one electron in its highest energy level. When this electron is lost, a lithium ion Li+ is formed.
Non metal ions
Non-metal ions:
- You need to be able to show the electronic structure of some common non-metal ions, using diagrams like these:
Chlorine, Cl
Chlorine is in Group 7. It has seven electrons in its highest energy level. It gains an electron from another atom in reactions, forming a chloride ion, Cl-.
ionic bonding
When metals react with non-metals, electrons are transferred from the metal atoms to the non-metal atoms, forming ions. The resulting compound is called an ionic compound.
Consider reactions between metals and non-metals, for example,
- sodium + chlorine → sodium chloride
- magnesium + oxygen → magnesium oxide
- calcium + chlorine → calcium chloride
In each of these reactions, the metal atoms give electrons to the non-metal atoms. The metal atoms become positive ions and the non-metal atoms become negative ions. There is a strong electrostatic force of attraction between these oppositely charged ions, called an ionic bond.
There are many ionic bonds in an ionic compound such as sodium chloride, arranged in giant lattice structures. Ionic compounds have high melting and boiling points.
Dot and Cross diagrams
You need to be able to draw dot-and-cross diagrams to show the ions in some common ionic compounds.
Sodium chloride, NaCl
Sodium ions have the formula Na+, while chloride ions have the formula Cl-. You need to show one sodium ion and one chloride ion. In the exam, make sure the dots and crosses are clear, but do not worry about colouring them.
Covalent bonding pt1
A covalent bond is a strong bond between two non-metal atoms. It consists of a shared pair of electrons. A covalent bond can be represented by a straight line or dot-and-cross diagram.
Hydrogen and chlorine can each form one covalent bond, oxygen two bonds, nitrogen three, while carbon can form four bonds
Covalent bonding pt2
Sharing electrons
A covalent bond forms when two non-metal atoms share a pair of electrons.
The electrons involved are in the highest occupied energy levels - or outer shells - of the atoms. An atom that shares one or more of its electrons will complete its highest occupied energy level.
Covalent bonds are strong - a lot of energy is needed to break them. Substances with covalent bonds often form molecules with low melting and boiling points, such as hydrogen and water.
Example - the animation shows a covalent bond being formed between a hydrogen atom and a chlorine atom, to form hydrogen chloride.
After bonding, the chlorine atom is now in contact with eight electrons in its highest energy level - so it is stable. The hydrogen atom is now in contact with two electrons in its highest energy level - so the hydrogen is also stable.
Representing covalent bonds pt1
Covalent bonds can be represented in several different ways.
- Straight lines and models: Straight lines are the most common way to represent covalent bonds, with each line representing a shared pair of electrons. 2-D or 3-D molecular models are especially useful for showing the relationship between atoms in multiple covalent bonds. Below are some examples of straight lines and images of 3-D models.
- Double and triple bonds Note that molecules can have a double covalent bond - meaning they have two shared pairs of electrons - or a triple covalent bond - three shared pairs of electrons. A double covalent bond is shown by a double line, and a triple bond by a triple line. A molecule of oxygen (O2) consists of two oxygen atoms held together by a double bond, like this:
A molecule of nitrogen (N2) has two nitrogen atoms held together by a triple bond, like this:
Representing covalent bonds pt2
Dot-and-cross diagrams
Dot-and-cross diagrams are another way to represent covalent bonds. The shared electron from one atom is shown as a dot, while the shared electron from the other atom is shown as a cross. Remember you only need to show the electrons in the highest energy level.
Hydrogen, H2
Hydrogen atoms can each form one covalent bond. One pair of electrons is shared in a hydrogen molecule (H2).
Representing covalent bonds pt3
You will also need to be able to draw dot-and-cross diagrams representing thecovalent bonds in the molecules of some common compounds:
Hydrogen chloride, HCl
Hydrogen atoms and chlorine atoms can each form one covalent bond. One pair ofelectrons is shared in a hydrogen chloride molecule (HCl).
Making polymers (Alkenes)
Hydrocarbons are compounds made from carbon and hydrogen atoms joined by covalent bonds. Alkanes are saturated - they have only single bonds. Alkenes have a double bond - they are unsaturated. Alkenes react with brown bromine water and decolourise it, but alkanes do not.
Alkenes can act as monomers. Under high pressure and in the presence of a catalyst many monomer molecules join together to make polymer molecules. These polymer molecules are saturated.
monomer- a molecule that can be bonded to other identical molecules to form a polymer
hydrocarbons and Alkanes pt1
Hydrocarbons
Most of the compounds in crude oil are hydrocarbons. This means they only contain hydrogen and carbon atoms, joined together by covalent bonds. Remember that a covalent bond is a shared pair of electrons.
Alkanes
The number of hydrogen atoms in an alkane is double the number of carbon atoms, plus two. For example, the molecular formula of methane is CH4. For ethane, it is C2H6.
Alkane molecules can be represented by displayed formulae in which each atom is shown as its symbol (C or H), and the covalent bonds between them by a straight line.
Structure of Alkanes
Alkanes are saturated hydrocarbons. This means their carbon atoms are joined to eachother by single covalent bonds.
Related discussions on The Student Room
- Maths for economists - constrained maximisation questoin »
- Edexcel GCSE Chemistry (Paper 1) »
- Edexcel Past Papers »
- Physics Question help »
- circle question help »
- best way to revise maths »
- How do I derive the order of growth in this code? »
- A level subjects for a bachelor in psychology yes or no? »
- Ln integration »
- Paramedic science undergraduate degree questions... »
Comments
Report
Report
Report