Biological Molecules 1: Water & Carbohydrates
- Created by: maylbrooke
- Created on: 12-09-17 08:58
Polymers
Polymers are made up of multiple monomors (single units) chemically bonded together.
They are formed by condensation reactions and broken up by hydrolysis reactions.
A condensation reaction is the joining together (polymerisation) of monomers producing water as a byproduct.
A hydrolysis reaction is the splitting of a bond ( to break down polymers into monomers) using water.
The equation for this is:
Monomer + Monomer + Monomer etc Polymer + Water
Carbohydrates
All carbohydrates are made up of only the three elements: Carbon (C) ,Oxygen (O) and Hydrogen (H).
The empirical formula for carbohydrates is Cn(H20)n
Carbohydrates are composed of monosacharrides which are a type of soluble and sweet monomer.
The number of carbon atoms in the monosaccharide tells you what type of sugar it is.
A monosaccharide with three carbons would be called a triose.
A monosaccharide with five carbons would be called a pentose.
A monosaccharide with six carbons would be called a hexose, eg. glucose.
Glucose
Glucose is a hexose sugar and a type of monosaccharide that forms hexagonal ring structures.
Glucose can come in different structural forms (isomers). These forms are called alpha glucose and beta glucose.
The difference between alpha and beta glucose is that the hydroxyl group (OH) on carbon 1 points downwards in alpha glucose whereas it points upwards in beta glucose.
The structural formula of alpha glucose is: The structural formula of beta glucose is:
Monosaccharides
Monosaccharides join together to form disaccharides (two monosaccharides) or polysaccharides (many monosaccharides).
When they join together they produce a molecule of water as a byproduct and form a glycosidic bond between the monosaccharides.
C6H12O6 + C6H12O6 = C12H22O11 + H20
Different monosaccharides product different disaccharides as the product of the condensation reaction.
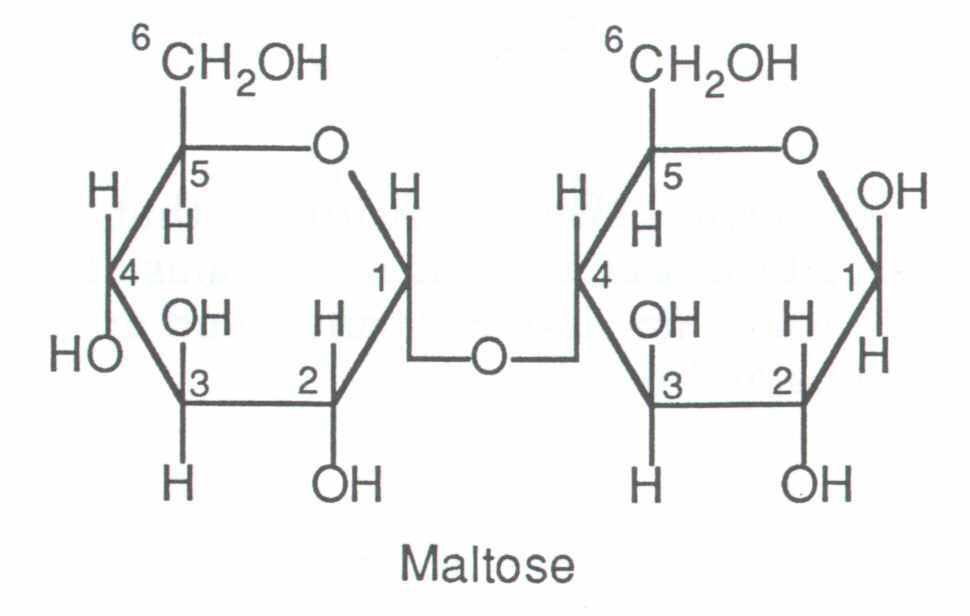
Maltose = Glucose + Glucose
Sucrose = Glucose + Fructose
Lactose = Glucose + Galactose
Condensation Reactions
When two monomers join together one of the the hydroxyl groups from each monosaccharide joins together to leave one oxygen and to produce a molecule of water.
The bond can be identified by the number of the carbon the hydroxyol is from on the first monomer and the carbon group it is from on the second monomer.
Eg two glucose molecules bond together to form maltose, which is connected by an alpha 1-4 glycosidic bond. It is shown as:
Starch and Glycogen
Starch is a polysaccharide made up of chains of alpha glucose and is found in plant cells.It is used for storage in plants and is composed of two different polymers: amylose and amylopectin.
Amylose is joined together in a compact helical shape as it forms only alpha 1-4 glycosidic bonds which allows it to store large amounts of glucose in a small space.
Amylopectin is branched as it forms alpha 1-4 and alpha 1-6 glycosidic bonds which allows the amylopectin to be quickly hydrolysed by enzymes to from maltose which can be used by the cell.
Glycogen is a polysaccharide also made up of chains of alpha glucose however it is found in animal cells. It is used for energy storage in animals and is found in liver cells and muscle tissue.
It is similar to amylopectin however it has more branches so it can be hydrolysed very quickly when needed.
Energy Storage Molecules
Both glycogen and starch molecules are very well suited for energy storage molecules. This is because:
- They are compact molecules so can store a large amount of glucose in a small space.
- They are large and insoluble molecules so cannot diffuse in or out of the cells.
- They have no osmotic effects as they are insoluble
- They are inert and so don't become involved in chemical reactions inside the cell.
- Glycogen and amylopectin (not amylose) can be quickly hydrolysed to soluble glucose due to their branched structure leaving many ends for the amylase enzymes to break down. This means they can be converted and used by the cells in which they are stored very easily.
Cellulose
Cellulose is made up of chains of beta glucose and is found in plant cells. It is formed by the condensation of beta glucose in which every alternate glucose molecule must be inverted hence it forming a straight chain.

It is a major component in plant cell walls because:
- It is made up of beta glucose which forms long straight unbranched chains.
- The chains run parallel to each other and therefore hydrogen bonds form crosslinks between them. As there are so many hydrogen bonds it makes the cellulose polymers collectively strong and have a high tensile strength. This is important as it ensures the cell wall stays rigid and prevents osmotic lysis.
- The cellulose molecules group together to from microfibrils which group to form fibres and provide more strength to the molecule.
Test for Starch
To test for starch you should:
- Add iodine solution to the sample.
- Record the colour changes.
The colour of the iodine solution (iodine in KI) is yellow to start with.
If the colour changes from yellow to blue-black then starch is present.
No heat is used in this test.
This is a qualitative test because although it tells you if starch is present or not it doesn't give you a value as to how much starch is in it. It is also objective as it relies on human observation of the colour change.
Test for Reducing Sugars
Reducing sugars are sugars with the ability to donate electrons. All the monosaccharides covered and some disaccharides are reducing sugars.
To test for reducing sugars you should:
- Add benedict's solution/reagent to the sample solution
- Heat the solution
- Record any colour change
If a reducing sugar is present then the colour should change from blue to orange-brown.
To test for non-reducing sugars you should:
- Test for reducing sugars, if the result is negative then continue
- Heat a new sample with dilute acid ( to hydrolyse the non-reducing sugar into its monosaccharides )
- Neutralise the solution with an alkali
- Test for reducing sugars again
If the second test is positive then the initial sample contained non-reducing sugars.
Problems with the Benedict's Test
There are several issues with the benedict's test which are:
- It is non-specific - It tells you if a reducing sugare is present but it does not tell you which one. A biosensor could be used to test for a specific sugar.
- It is qualitative / semi-quantitative - The result is determined by colour change so you cannot measure an exact value for it. It can be classed as semi-quantitative as you can get an estimate of the amount of sugar present depending on the colour change.
( Blue - None, Green - Very low, Yellow - Low, Orange-Brown - Medium, Red - High)
- It is subjective - It relies on the individuals perception of the colour change to get a result. A colorimeter can be used to make it objective and non-biased, as a colorimeter can quantify the results.
A colorimeter can be used to measure an exact value for the solution. A serial dilution of glucose solution can be carried out producing a set of solutions with known concentrations. The benedict's test can then be performed on each of these and the colour intensity measured using a colorimeter. This then allows you to produce a calibration curve which can be used to read the concentration of glucose in an unknown solution.
Inorganic Ions
An ion is a charged atom ( an atom that has lost or gained electrons ). Inorganic ions occur in solution in the cytoplasm of cells and in body fluids ( eg. blood or tissue fluids ).
Some common ions are: Iron ions (cation), Sodium ions (cation), Phosphate ions (anion), Hydrogen ions (cation).
Each of these ions has a use in the body. These uses are:
- Hydrogen Ions - Affects PH levels
- Iron Ions - Component of haemoglobin in red blood cells
- Phosphate Ions - Component of DNA, ATP, and phospholipids
- Sodium Ions - Involved in the absorption of glucose and amino acids in the small intestine by the process of cotransport.
Water - Dipolar Molecule
A water molecule is made up of two hydrogens atoms and one oxygen atoms. Each hydrogen atom is slightly positively charged whereas the oxygen atom is slightly negatively charged. These charges cancel out overall making the atom neutral.
The oxygen atom is at one end of the water molecule and the hydrogen atoms are at the other end which causes the end with the oxygen to be slightly negatively charged (delta - ) and the end with the two hydrogen atoms to be slightly positively charged ( delta + ).
For this reason water is a dipolar molecule as it has both negative and positive poles.
Hydrogen Bonding in Water
The positive pole of one water molecule is attracted to the negative pole of another water molecule. This attractive force between teh oppositely charged poles is called a hydrogen bond.
A hydrogen bond is relatively weak as its only about 1/10 the strength of a covalent bond.
In water however there are massive numbers of hydrogen bonds between many water molecules therefore the overall strength of all the hydrogen bonds combined is quite high.
The hydrogen bonds are resposible for some of the unusual properties in water such as the specific heat capacity it has and the latent heat of vaporisation.
Specific Heat Capacity & Latent Heat
The specific heat capacity of a substance is the amount of energy needed to change the temperature of 1kg of a substance by 1 degrees celcius.Different substances have different specific heat capacities depending on their properties.
Water has a high specific capacity ( the highest of any common liquid ) due to the hydrogen bonding in water. As there are so many hydrogen bonds between water molecules they stick together and so a lot of energy is required to break those bonds. That is also why it has a higher boiling point that might have been otherwise predicted.
In order for a substance to change from a liquid to a gas, energy is required to break the forces between the molecules. The latent heat of vaporisation is the amount of heat (energy) required to evaporate 1g of water.
The large amount of hydrogen bonds means that a large amount of energy is required to change states and it therefore has a high latent heat of vaporisation. This property is the reason sweating is such an effective cooling technique as the water molecules produced take a large amount of heat energy to evaporate so they remove a lot of heat energy from the surface of the skin cooling it.
Cohesion and Surface Tension of Water
Cohesion is when molecules of the same substance stick together.
The large amounts of hydrogen bonds in water cause it to have strong cohesive forces. This allows it to be pulled up through a tube like a pipette, straw or the xylem tissue in plants.
This also means that when water meets air the water molecules are pulled back into the body of water rather than escaping into the air. This force is called surface tension.
Surface tension means the water acts like a skin and requires force to be broken.
Certain organisms make use of this and can walk on or be supported by the water's surface, eg. pond skaters.
Water's Functions in Living Organisms
Water is a major component in all living organisms and is essential to life. It is one of the raw materials used in photosynthesis and is a product of respiration.
Water is the medium in which enzyme-catalysed reactions take place.
Water is also a solvent and dissolves substances such as:
- respiratory gases (oxygen and carbon dioxide)
- excretory products (ammonia and urea)
- inorganic ions (hydrogen, iron, sodium, chloride and phosphate)
- small hydrophilic molecules (amino acids, monosaccharides, ATP)
Water is used in hydrolysis reactions to break down complex molecules ( eg. proteins to amino acids). It is formed in condensation reactions (eg. starch and protein synthesis)
Related discussions on The Student Room
- Do I need to know how to draw structures for carbohydrates? (AQA A Level Bio) »
- Paper 3 AQA a Level biology »
- Mark my AS level bio essay pls »
- Any good youtube channels for Bio + Chem a levels? »
- exams 2022 »
- 25 mark essay question »
- Animal Transport Notes OCR A Level »
- How to answer 6-mark questions in A-Level Biology? »
- Calvin Cycle »
- Alevel biology understanding »
Comments
No comments have yet been made