Applied science - Physics
- Created by: Breedelahaye
- Created on: 12-01-20 13:30
Thermal energy
What is thermal energy?
Thermal energy is the energy that you cannot see or measure that is stored in their vibrations, rotations or other random moverments.
Random molecular motion of thermal energy
The kinetic theory of matter describes;
- Solids as atoms having fixed positions but able to vibrate.
- Liquids as molecules flowing past on another and colliding, while inside the molecule atoms still vibrate.
- Gases as moleculles separated and travelling fast between collisions. The atoms still vibrate and the molecules can spin.
- Thermal enrergy is stored in these motions.
- It is added to whenever work is done (electric currents pass through them, drilled, stirred, sawn.
Heat flow. Q of thermal energy
If two bodies are in thermal contract:
- The quantity of thermal energy that is transferred between the two bodies is called heat
- Heat transfer happens by conduction, convection and raditaion
- Heat flows from the hotter to the cooler, and will keep flowing until they reach thermal equilibrium (Reaches the same temperature).
Temperature, T of thermal energy
The Zeroth law of Thermodynamics states:
- There exists a physical temperature, such that
- Any two bodies at the same temperaturre will be in thermal equilibrium with one another.
- A thermomometer can be anything with a measurable ohysicaln property that varies directly with its temperature.
- A thermometer has to come to thermal equilibrium with its surroundings before it can be read.
Change of state (Thermal energy)
Heat transfer into a material will cause;
- Either temperature change: sensible heat (that is, you can sense its effect)
- A change of state (solids to liquid to gas/vapour, or vice versa) ; latent heat (latent means lying hidden as binding energy of the material - you sense no temerpature change)
- During a change of state, latent heat is either absorbed or given out without a temperature change.
Energy and work
To do work you need to transfer energy. It might be from potential energy, kinetic energy or it can be from other types of energy.
Force, F and displacement, S
- Force and displacemet (distance moved from a reference point) are vectors - that is they each have a direction as well as a size.
- Direction matters - the force and the distance must be in the same direction.
- Work done = force x distance moved in the same direction
Other facts for energy and work
Gravitational potential energy = force of gravity (mg) x vertical distance moved (triangle h)
Kinetic energy = 1/2mv2
(Velocity, v, is the result of acceleration, a, caused by a force, F = ma)
Pressure, p and volume V, of energy and work
- A liquid or gas exerts pressure in all directions.
- When it changes volume by flowing or expanding it does work, pushing against its surroundings.
- Work done = pressure x volume change
- W= P triange V
- Pressure is measured in Pascal (Pa), which is the same as Newton per square metre (Nm-2)
- Volume is in cubic metres (m3)
- f
- Remember to convert units:
- 1 m3 = 1000 litres
- 106 cm3
- 109 mm3
Power, Energy and work
- Power is the rate of using energy or doing work.
- The uunit for energy is joule (J)
- The unit for power is watt (W) =J/s
- f
- You need to recoignise the following big multiples:
- Kilowatt (kW) = 1000 watts
- Megawatt (MW) = 106 Watts
- Gigawatt (GW) = 109 Watt
Hydraulic braking system for energy and work

Other forms of energy
- Chemical energy (chemical bonding)
- Nuclear energy (nuclear reactions)
- Electrical energy
- Thermal energy (associated with temperature)
Gases and thermometry
To make valid measurements with a thermometer you first need to calibarate it against a temperature scale, but to establish the scale you need a chosen standard thermometer.
Ideal gases - pressure and volume. Gases and therm
The ideal gas equation, pV = NkT, comes from kinetic theroy, where we assume that gas molecules behave like;
- point particles, with
- elastic collisions, and
- no significant binding forces between them.
N is the number of particles. K is Boltzmann's constant, 1.38 x 10- 23 J/K.
T in this equation is proportional to the average kinetic enrgy of the gas molecules - an ideal concept of temperature.
So, an ideal gas would make a perfect standard thermometer.
Real gases behave very like an ideal gas when they are well away from their condensation point - higher temperature, lower pressure.
Absolute zero - Gases and thermometry
- O Kelvin (K) = -273.15 degree celcius
- Is where all themal energy has been removed.
- All molecules are in their ground (lowest possible energy) states
- Cannot be practically achieved, though we can get close to it.

Practical thermometres you might use
Thermometer type = As temperature rises
Liquid in glass = Liquid expands
Thermistor = Electrical resistance decreses
Platinium resistance = electrical resistance increases
Infrared = increase in IR raditaion emitted
Thermocouple = voltage diffrence increases
Energy conservation
A helpful in thermal physics or chemistry is draw an imaginary boundary line around each system that you are investigating, and to measure the transfer of energy in and out.
Energy transfers - work and heat (Energy conservat
The system might be: hot gases inside an engine; a chemical reaction chamber; a single living cell or a whole ecosystem.
- Heat transfers, Q, into the stystem count as positive; flows out count as negative.
- Work done by the system (hot gases exanding) counts as positive.
- The actual direction of heat tranfers depend on the temperatures outside the system - in external temperature reservoirs, which might be specially designed heat exchangers or just the system's surroundings.
Internal energy, U (Energy conservation)
Systems are complex, alaways with huge numbers of moleculars and oftern with many parts. The internal energy of a system includes all its:
- thermal energy (heat capacity)
- chemical and nuclear bond energies
- energy associated with physical states of matter (latent heat)
- kinetic or potential energy
- So U is far too complex to ever measure in total. We only calculate its changes;
- Triangle U = U2 -U1
The First Law of Thermodynamics
This is just a statement of the Principle of Conservation of Energy as it applies to a thermal system:
Q = Triangle U + W
Heat in = emergy change in the system + work out
- or in the case of the system shown above with two heat reservoirs at diffrent temperatures:
Q(in) - Q(out) = triangle U + work
Specific heat capacity, c
The heat required to increase the temperarure of 1 kg by 1kelvin (or degree celcius)
Q = mc triangle
For water:
C = 4.18 KJKG-1 K-1
Specific latent heat, L
The heat required to chnage the physical state of 1kg of a material
Q = mL
L has seoarete values for:
- Fusion (melting) - For ice to water: L = 333.6 kJ kg-1
- Vaporisation - For water to steam: L = 2,26 MJkg-1
Processes
To change a system from one state (that is, a specific pressure, temperature, volume, internal energy) to another there are many possible diffrent process paths that could be followed and each involves iffrent amounts of work and of heat transfer.
Natural processes
As time progesses, some things tend naturally to occur. For instance:
- Water flows downhill
- Heat flows from a hotter body to a colder one
- Substances mix
- Gases expand to fill the space available
- Exoothernic chemical reactions
- Friction turns mechaninical work into heat
The reverse of these processes never occurs naturally - they are irreversible.
The Second Law of Thermodynamics
There are several equivalent ways of stating this law
- A natural process can never be reserved in its entirely.
- It is impossible to completly change heat into work.
- Heat will not flow from a colder body to a hotter one without an input of work.
- The entropy of the universe is always increasing.
- You cannot reverse the direction of time.
Reversible processes
Reversible processes are an ideal, as there are no fully reversible processes. It is a useful idea of thinking about equilibrium and about the limits of efficiency. There are two possible types of reversible process: adiabatic and isothermal.
Adiabatic (thermally isolated) processes
There is no heat transfer, Q = O
- Work input = internal energy gained, -W = triangle U, that is, no energy is wasted.
- Compressing a gas adiabatically leads to a corresponding temperature rise.
- Real systems, even if well-insulated, are never fully insulated thermally.
- Changing things too quickly for heat to be lost can be near adiabatic, but friction and viscous drag will mean it is still not fully reversible.
Isothermal
Processes
- Two bodies in thermal equilibrium (that is, at the same 7)
- Heat can flow back and forth in either direction reversibily.
- But with zero temperature diffrences there would be no net heat transfer.
- Real systems with very small temperature diffrence, for example, heat exchanges, can be almost reversible. They are large and slow.
Cycles
Engines, refrigerators and living organisms all use cycles of processes to cause changes and energy transfer to/from their surroundings while the system itself remains unchanged overall.
Ideal reversible (Carnot) cycle
Comprises:
- Two idothermal processes at diffrent temperatures, TH (hot) and Tc (Cold) with heat transfer amounts, Q in and Q out.
- Two adiabatic processes - where Q = O by definition
- On a pressure - volume diagram, the area under the p-V curve for each process is the work done. Thus, the net work done in the cycle, W, is the area enclosed - positive when the direction of the cycle is clockwise, that is, for a heat engine.
Heat engines - internal combustion
Power station steam turbines
- Comes quite close to the ideal cycle by using very large, heavy heat exchangers.
Intwenal combustion engines
- Save the huge weight of a boiler heat exchanger by burning the fuell inside the cylinder where it then expands, doing work.
- Benefit from a higher thermal efficiency due to the very high gas temperature produced by fuel/air compression followed by combustion.
Efficiency
How much you usefully get out for the energy that you put in? When the unput enegry is heat then there are limits to what you can do with it:
efficiency = useful energy output / total energy input
Useful and wasted energy
- With engines, heat counts as wasted if it cannot be converted into useful work.
- Heated buildings, the waste heat from an engine can become useful heating.
- Example combined heat and power scheme.
- Escaped heat would then be wasted.
Maximum efficiency (Carnot) engines
- No engines can be more thermally efficient than an ideal reversible Carnot cycle engine, for which:
- Qout / Qin = Tc/Th
Moving heat
Cooling or heating spaces to the desired temperature is becoming the biggest area of energy management. Reverse cycle vapour compression does this extremely cost effectively.
Refrigerators and heat pumps
Both use the same basic equipment, but:
- Refridgerators cool an enclosed space and reject heat outside.
- Heat pumps draw in heat from the ground or outside air and move that heat into a room or hot water tank at a higher temperature.
So they are distinguished by:
- The positioning of heat exchangers, and
- Which space is being temperature controlled
Basic refrigeration
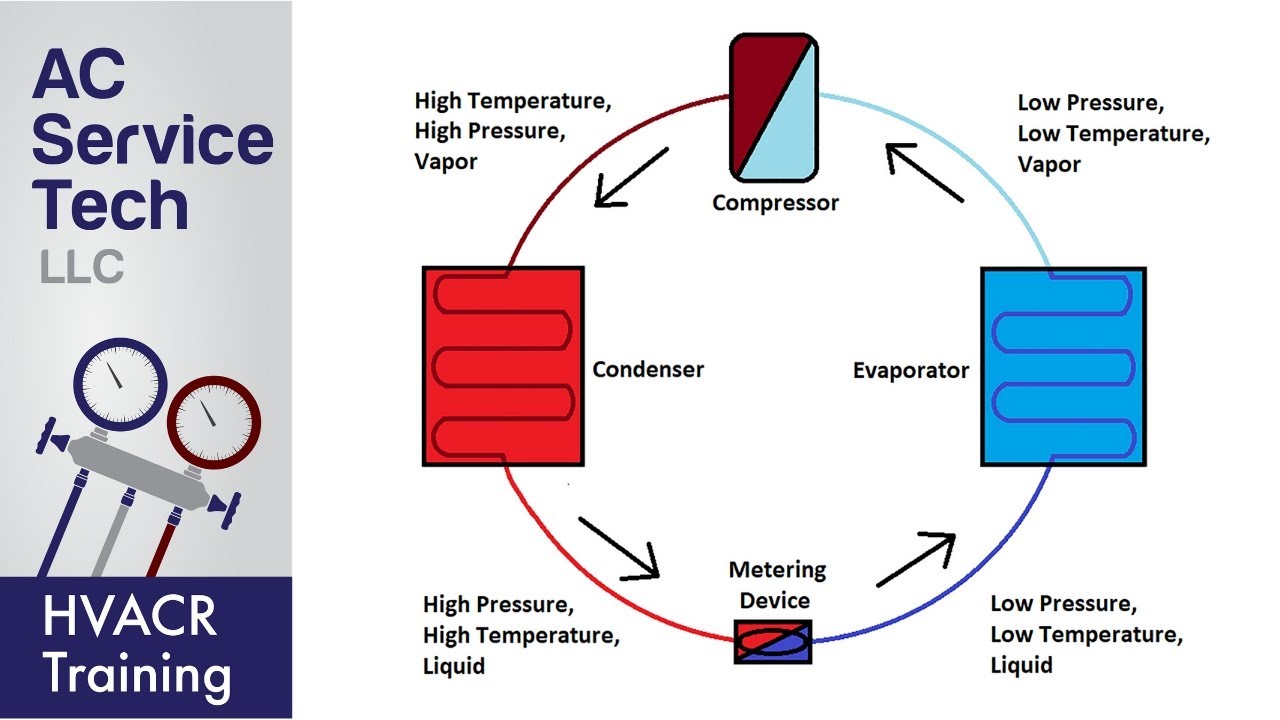
Coefficient of performance
CoP is the inverse of thermal efficiency
-> number bigger than 1
- Heat moved, Q is what is useful
- Work input, W, is the cost.
- So, maximum theoretical (reversible Carnot cycle) CoP values are:
- For refrigeration: Qin/W =Tc/Th-Tc)
- For heat pumps: Qout/W=Th/(Th-Tc)
Common refrigerants
- Chlorofluorocarbons (CFCS) - but they damage the ozone layer.
- Alkanes - still some air pollution
- Ammonia - needs large equipment
- Carbon dioxide - no latent heat above critical point of 31 degree celcius.
To improve actual CoP value
You should try to:
- Minimise temperature gap (Th -Tc) so:
- Use oversize heat exchangers.
- Tap large steady heat sources like underground water.
- Expand a liquid because the volume change is small, so not much work is lost.
- Cut friction or viscous flow losses.
- Keep compression work low - gas heat capacities affect this.
Choice of working fluid
- High latent heat of vaporisation/condensation helps by keeping heat transfer processes isothermal and near reversible.
- This is why water/steam is a good fluid for heat engines.
- Refrigerators and heat pumps, using denser vapours keep machinery more compact.
- Boiling and freezing points of refrigerants need to match with system temperature.
Elasticity
Elastic behaviour is the ability of a material to spring back to its original shape and size after being stretched, squashed or otherwise distorted.
Hooke's law
Force, F, is proportional to extension, DeltaX
F = K delta X
Where K is a constant.
This is:
- About stretching a material (tension) or sqeezing it (compression)
- Not a general law
- Obeyed by metals and some other materials for a limited range - that is, up to the limit of proportionality.
Tensile/compression stress and strain
These enable you to take into account the dimension of the origanal sample:
stress = force/cross-section area
o = F/A (Units: N m-2 or Pa)
Young's modulus (elastic module), E
E = stress/strain N m-2 or Pa
Rearranging this gives another way of expressing Hooke's law
Elastic limit (Yield strength)
- Highest tensile stress with full elastic recovery
- Beyond this the material yields to give a permanent plastic deformation.
- Hooke's law and Young's modulus fail to apply from shortly before this point.
String and elastic energy
This work done (energy stored) is equal to the area under the F vs Triangle X graph
W = 1/2 F delta x
= 1/2 K triangle x 2
Elastic hysteresis
Elastic hysteresis occurs in materials like rubber, due to internal friction:
- Both stretching and recovery take time.
- Work turns to heat.
They absorb energyt these are useful materials for cushioning shock and for damping of oscillations.
Shape change
Plastic means able to be formed. It describes materials expanding a permanent change in shape without completely breaking up, and happens after they pass the elastic limit.
Plastic deformation
- When stress exceeds the yield strength, the Crystalline structure of metal allows layers of atoms to glide over each other.
- When stress is removed the material stays deformed - has a permanent set.
- Amount of plasticity depends on the microcrystalline structure of the metal - so varies between meterials.
- Most non-metals show little or no plasticity, some plastics polymers materials for example, nylon, can behave rather similarly to metals.
Ductility
Ductility is ability to be shaped by plastic flow under tension:
- drawing into rods, wire or tubes
- Deep drawing of sheet material into bowls, cans, etc
Ductility depwnds on temperature. If cooled sufficiency even metals become brittle.
Malleability
Mallebility is ability to be shaped by plastic flow under compression
- rolling or hammering into sheets
- cold forming by stamping or pressing
Some metals are malleable but not ductile.
Many plastics materials and amorpous solids like clay are malleable.
Stainless steel
- Produced by drawing through a die to form thin walls - a tensile process that requires ductility.
Steel screws
Produced by drop forging - a compressive process with a hammer and die on either hot or cold metal, which requires malleability.
Lead sheet
Is produced by hammering and rolling - compressive processes that depend on malleability.
Failure
Strength
- Ultimate tensile strength is the highest stress a material can sustaine, just before it breaks.
- Ductile materials, there is a lower yield strength - the stress at which plastic deformation starts.
- Brittle materials like concrete or g;*** only have a UTS - no yield point, no plastic deformation.
Brittle strength
- Happens when stress becomes concreated at the tip of tiny inoerfections and cracks that exist in the material.
- Cracks grow rapidly (visable under X-rays) and break right across the iteam.
- Most ductile material harden as they are worked until stress levels become high enough to cause a brittle failure.
Creep
- Creep is a slow version of plastic deformation.
- It increases with temperature, and could eventually lead to failure if components no longer fit or if they neck in.
Fluid flow
Moving of liquids or gases differs from moving solids - because fluids do not have a fixed shape - but it is still above driving forces, resistance and energy used.
Streamline flow
Also called a laminar flow
- Occurs at lower values of flow rate and pressure diffrences.
- Drift velocities of particles are all parallel and in the same sense.
- Fluid in contract with a solid surface has virtually the same velocity as that of the surface.
- Velocity changes across the flow of the stream
- It is the most energy efficient type of flow - so it is what engineers usually aim for unless mixing is needed.
Viscosity
Fluids vary widely in their resistance to flow - diffrent visosities
- Layers of fluid moving at diffrent speeds cause a velocity gradient.
- Dynamic visocity defined by Newton's law of viscosity using a simple streamlibe flow model.
Non-Newtonian fluid
This is a fluid that is a constant calue at any given temperature and pressure. But non-Newtonian, whose viscosities change in other ways, have useful applications.
Shear thinning and shear thickening fluids
These fluids change viscoity as soon as there is a shear stress
- Mostly these are colloidal suspensions of solid particals or droplets in a liquid.
- Brusjong, sliding or stirring a liquid provides shear stress.
Time-dependent behaviour
- Thixotropic fluids thin gradually on strirring then slowly reset.
- Rheopectic behaviour is rare - a time dependent thickening with shear stress, thinning again when it stops
Bingham plastics
- Behave like solids at low shear stresses, but above a yield stress they begin to flow, e,g toothpaste
Fluid dynamics
Mass flow continuity
Once a steady state flow has been established in a system of pipes, mass flow rate must be the same;
- Entering the system
- Leaving from its outlet
- Crossing every boundary along its length
Mass flow continuity means that you can measure mass slow at any poing that you choose along the flow.
Pressure drop in pipes
- Pressure drop increases if the viscosity, increases and if there is turbulence.
- Pipe radius has a dramatic effect - pressure drop varies inversely with the 4th power of r.
Air-plane wing
![Image result for Diagram of airplane wing]](https://cdn4.explainthatstuff.com/how-airfoil-wing-makes-lift.png)
Related discussions on The Student Room
- Universities »
- Does geography count as another science? »
- Cambridge »
- Official NATURAL SCIENCES applicants thread 2024 »
- Personal Statement for science courses »
- Natural science advice?? »
- What a-levels should be the best for computer science degree? »
- Can i do radiographer degree with health and social care btect level 3 »
- Natural science course »
- NatSci at Cambridge »

Comments
No comments have yet been made